What is cell Culture?
Cell culture is the process of growing cells in a controlled environment outside of their natural context. This is done by removing cells from animal or plant tissue, breaking them apart, and cultivating them in an artificial environment. Cell culture is used to study the behaviour and characteristics of cells, and is an important tool in fields such as medicine, genetics, and biotechnology.
Limiting Factors with Scaled up Cell culture
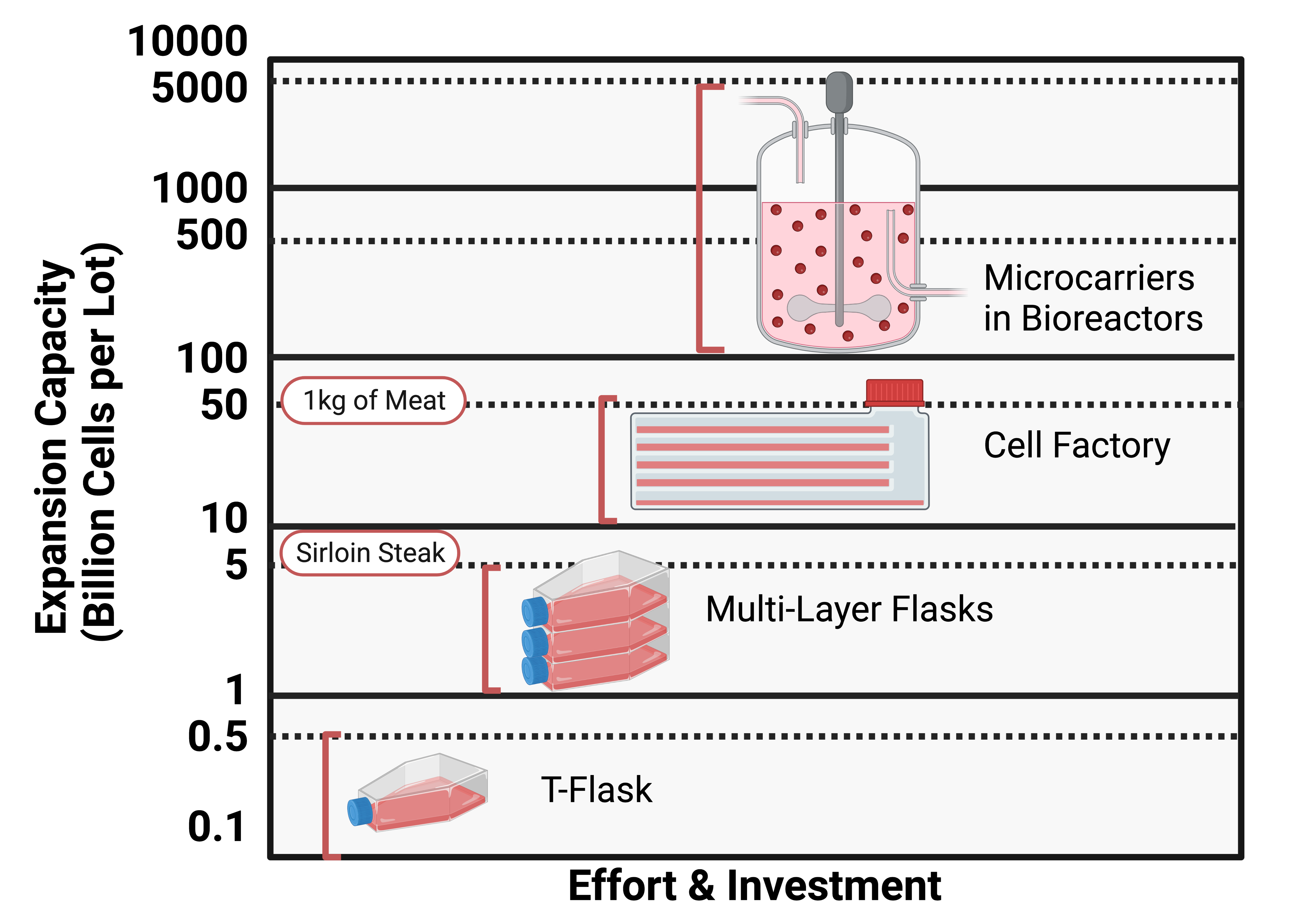
Scaling up cell culture can be challenging due to the limited surface area for cell growth. Traditional small-scale methods, such as plastic flasks, become impractical when attempting to grow cells on a large scale. Technologies like multi-layer flasks and cell factories can improve the surface area-to-volume ratio, but they are not scalable solutions. Bioreactors, which are large stirring tanks, offer a more efficient and automated solution for cell culture. However, these are not suitable for growing cells that require a substrate, such as adherent cells. To overcome this limitation, microcarriers have been developed to allow the growth of these types of cells in suspension in a bioreactor. Despite the challenges, advances in technology and research are helping to mitigate limitations and enable successful large-scale cell culture.
Microcarrier
Microcarriers are small, spherical support matrices that allow for the growth of adherent cells in bioreactors. By culturing cells on the surface of these microcarriers, each particle carries several hundred cells, which greatly increases the expansion capacity of the culture system. This makes microcarriers a valuable tool for scaling up culture systems for industrial production of cell or protein-based therapies, or for research purposes. They provide a simple and efficient way to grow large numbers of cells in a controlled, suspended environment, making them an important innovation in the field of cell culture.
Issues with Microcarriers
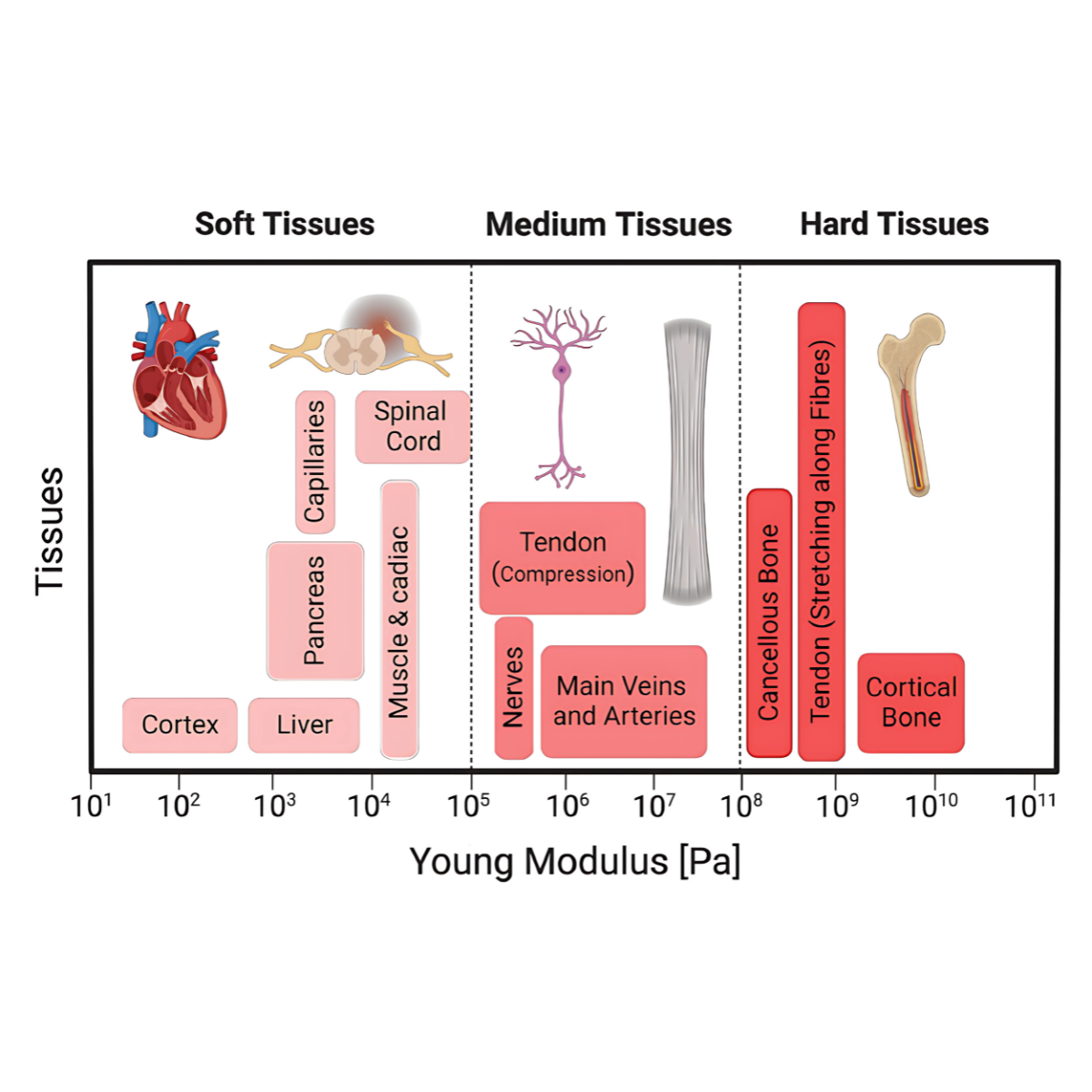
Current microcarriers face several challenges across different applications. They can be costly, have cell loss rates of more than 50%, and are often made from non-edible materials like plastic or glass, which can leave particulates in the final product. Different cell types also have very different growth requirements, some, like osteoblasts, form rigid mineralised tissue, while others, like muscle or fat cells, are much softer and more delicate. A one-size-fits-all approach to microcarrier design often leads to poor cell attachment, reduced viability, and lower yields. This highlights the need for customisable microcarriers that are cost-effective, safe, and specifically tailored to the requirements of each cell type, improving performance and scalability in a wide range of bioprocesses.