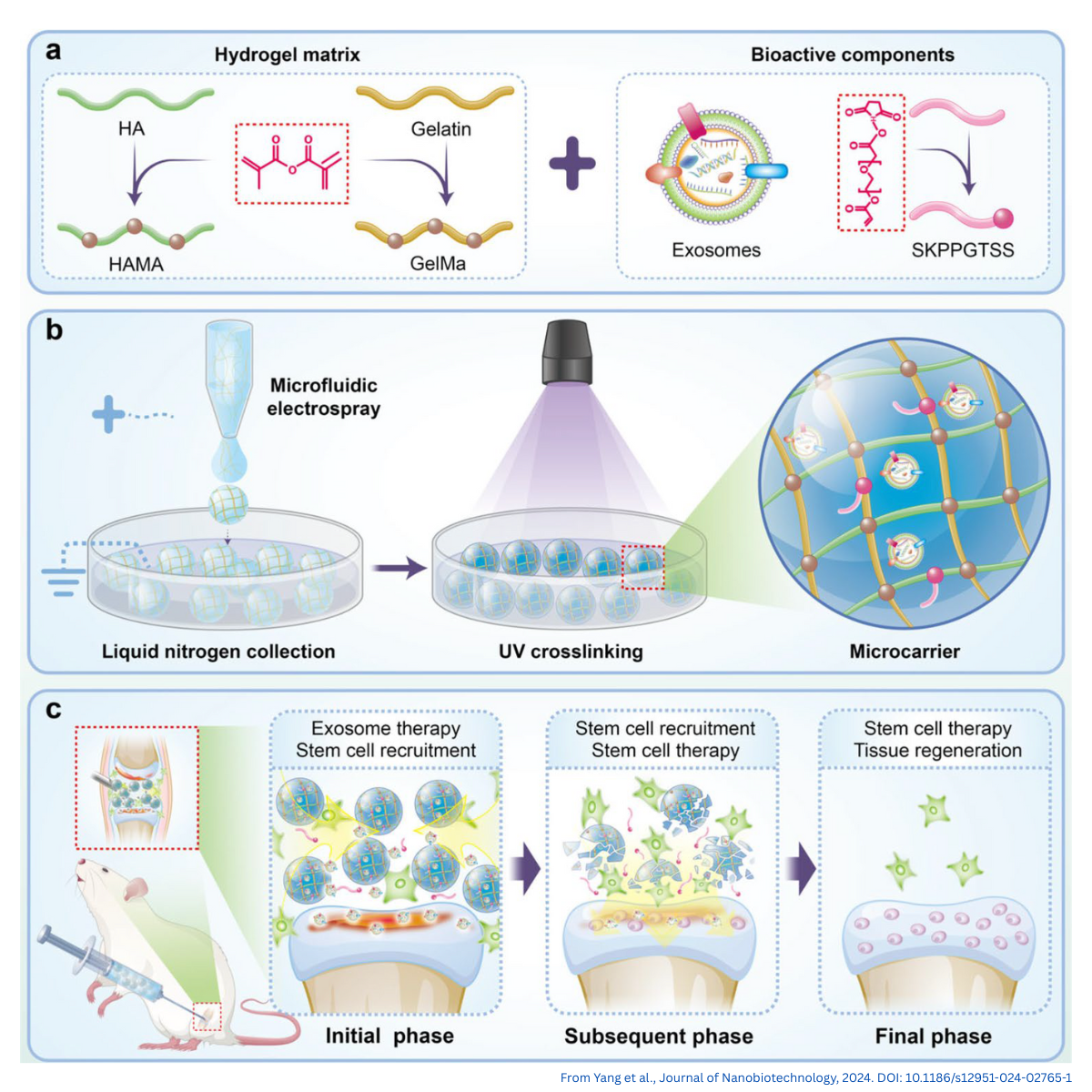
How to Expedite Microcarrier Dissolution While Maintaining Cell Quality
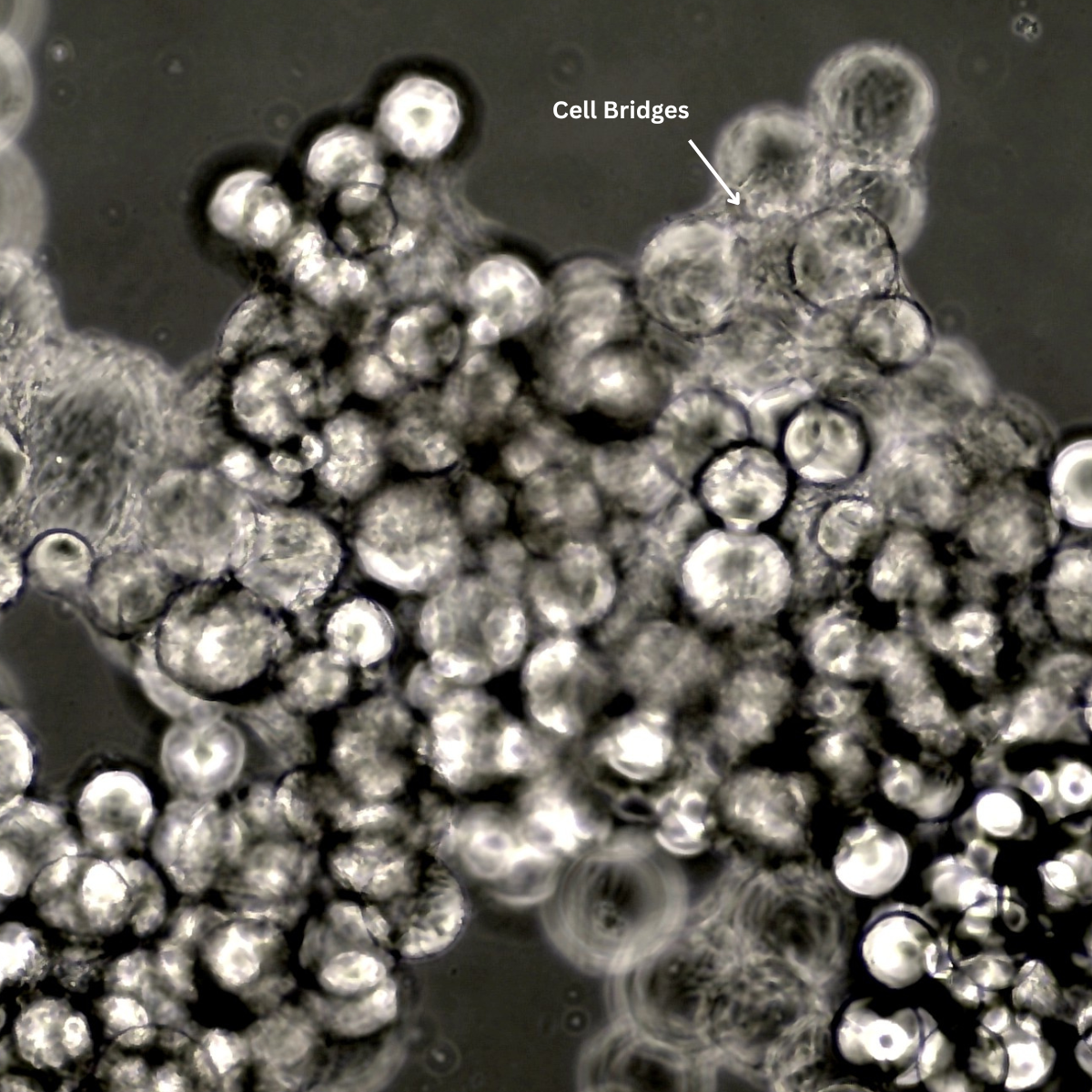
“Why is microcarrier dissolution taking longer than expected?” Microcarrier dissolution is a key step during harvest, but delays are usually linked to how the process is handled. In our experience, when the workflow is well understood and the steps are followed properly, dissolution is straightforward. When something isn’t quite right, or the process is [...]