A closer look at the research by Díaz-Maneh et al. (2026)
The Problem: Scaling Fat Production for Cultured Meat
When we talk about the future of food, cultured meat is often at the centre of the conversation. But there’s a secret ingredient that makes meat truly delicious: fat. It provides the flavour, the juicy texture, and that unmistakable “sizzle” when it hits the pan.
The big question for scientists is: How do we grow enough fat to feed the world?
A key challenge in cultured meat production is scaling fat cells efficiently while maintaining high cell viability, density, and functionality.
A recent study by Díaz-Maneh et al. (2026) offers a fascinating case study on how tiny beads called microcarriers are the key to solving this “scaling” problem using duck cells.
The Problem: Cells Need a Place to Sit
Most animal cells, including those that become duck fat, are “anchorage-dependent.” This means they can’t just float around in a tank; they need a solid surface to grab onto so they can grow and multiply.
In a small lab flask, this is easy. But when you try to scale up to a giant bioreactor (essentially a large, high-tech fermentation tank), providing enough surface area becomes a massive challenge.
This limitation is one of the main bottlenecks in large-scale adherent cell culture and cellular agriculture processes.
The Solution: Microcarriers
Microcarriers are microscopic beads that act as a “movable floor” for cells. By adding these beads into a bioreactor, we can:
- Massively increase the surface area without needing a bigger tank.
- Keep cells healthy while they are being gently stirred.
- Grow millions of cells in a relatively small space.
This makes microcarriers a key enabling technology for scalable bioreactor-based cell culture systems.
In this study, microcarriers were used as a platform to support both cell expansion and subsequent fat production.
3 Key Lessons from the Duck Fat Study
1. It’s All About the “Grip”
The researchers found that over 90% of the duck cells successfully attached to the microcarriers. This is a huge win, it proves that these cells are very happy to grow on beads, making the expansion process much more efficient.
Cells showed rapid attachment to microcarriers, with approximately 70% attachment at 7 minutes post-seeding and reaching 93.6% by 14 minutes, indicating highly efficient and reproducible cell seeding.
This strong attachment supports higher cell densities and improves overall process scalability.
2. Beware of the “Whirlpool” (Shear Stress)
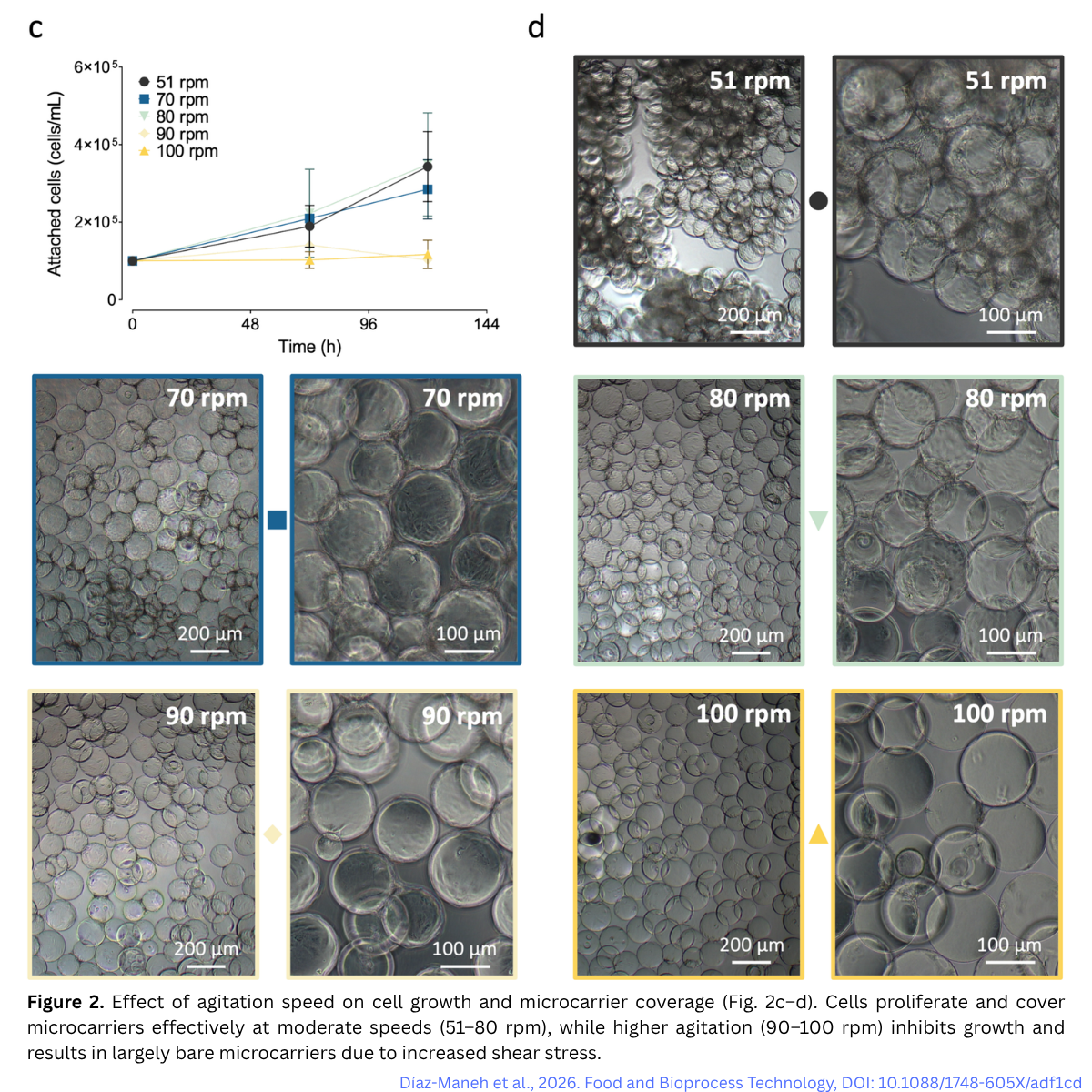
One of the trickiest parts of using a bioreactor is the movement of the liquid. If you stir too fast, the “mixing forces” (called shear stress) can actually peel the cells right off the beads or damage them.
The finding: the cells were quite sensitive. The team had to find the “Goldilocks” zone, stirring fast enough to keep oxygen moving, but gentle enough to keep the cells safe.
In shake flask experiments, cell growth was maintained within an agitation range of 51 to 80 rpm. Under these conditions, cells reached approximately 200,000 cells/mL by 72 hours, and up to 425,000 cells/mL at 120 hours with medium replacement. In contrast, cells failed to proliferate at 90 and 100 rpm, indicating that these conditions exceeded the cells’ shear tolerance.
When the process was scaled up, the same principle held true. In a stirred tank bioreactor, a low agitation speed of 50 rpm was used, just enough to keep the microcarriers suspended while minimising stress. Under these conditions, cells successfully expanded to approximately 489,000 cells/mL, with near-complete attachment maintained throughout the culture.
This highlights a key lesson: microcarrier cultures require careful control of shear forces, especially when working with sensitive primary cells.
3. From Growth to Gourmet Fat

It’s not enough just to grow the cells; you have to turn them into fat. After the cells multiplied on the microcarriers, the team used a special “fat-making” medium.
The result: the cells successfully filled up with lipid droplets (real fat). In shake flask optimisation studies, cells reached a specific triglyceride accumulation of 314 pg per cell under optimised differentiation conditions.
At bioreactor scale, adipogenesis was successfully reproduced. Following induction at around 400,000 cells/mL, cells continued to accumulate lipids, reaching approximately 380 pg per cell by the end of the culture.
In terms of cell expansion, the stirred tank bioreactor supported growth up to 489,000 cells/mL, demonstrating scalability from shake flask conditions.
In addition, optimised feeding strategies were critical. A cell-specific perfusion rate (CSPR) of ~0.779 nL·cell⁻¹·day⁻¹ was identified as the minimum required to sustain proliferation, while differentiation was optimised at 1.83 nL·cell⁻¹·day⁻¹, ensuring both cell survival and lipid accumulation.
This shows that microcarriers can support both high-density cell expansion and functional fat production.
Why the “Stirred Tank” Won
The study compared different types of bioreactors, including rocking motion bioreactors (RMB) and orbitally shaken bioreactors (OSB), and found that the Stirred Tank Bioreactor (STB), which uses an impeller-based mixing system, was the most effective.
Both RMB and OSB systems failed to support sustained cell growth. In these systems, cells either died within a few days or failed to proliferate, even when agitation conditions were reduced to minimise shear stress.
In contrast, the STB supported robust cell growth, maintained high cell viability, and enabled successful fat production. Cell densities reached nearly 489,000 cells per mL, with close to 100% cell attachment maintained throughout the culture.
Importantly, the STB was the only system that successfully supported both cell proliferation and adipogenic differentiation, with triglyceride accumulation reaching levels comparable to those observed in shake flask cultures.
This reinforces that stirred tank systems are well-suited for scalable microcarrier-based manufacturing processes, particularly when working with shear-sensitive primary cells.
The Simple Takeaway
This research is a proof of concept that cultured fat can be produced at scale. It shows that by pairing the right microcarriers with the right bioreactor settings, it is possible to create key components of meat in a controlled, lab-based environment.
Importantly, it demonstrates that microcarriers enable:
- high cell attachment efficiency
- high-density cell culture
- scalable bioreactor integration
- functional differentiation into fat cells
As the industry moves forward, the focus is shifting toward improving microcarrier systems, developing materials that are more cost-effective, application-specific, and better suited for sensitive cell types.
This is an area of active innovation across the field, including efforts to design more cell-friendly and scalable microcarrier platforms for applications in cellular agriculture.
Reference:
Díaz-Maneh, A., Camacho-Sucarrats, A., Fuenmayor, J., Ilic, N., García-Aranda, N., Enciso, Y., Notari, M., Gòdia, F., Lavado-García, J., & Revilla-Sánchez, R. (2026). Proof-of-Concept for Cell Culture-Based Duck Fat Production on a Laboratory Scale. Food and Bioprocess Technology, 19(4). https://doi.org/10.1007/s11947-026-04259-4
Disclaimer
Smart MCs is not affiliated with the authors of this work. This study did not use Smart MCs microcarriers. This post is intended to highlight general use cases of microcarriers in the industry for educational purposes.
About Smart MCs Microcarriers
While this study did not use Smart MCs microcarriers, it highlights many of the key challenges our materials are designed to address.
At Smart MCs, we develop microcarrier platforms focused on supporting:
- reliable cell attachment and expansion
- scalability in bioreactor systems
- compatibility with sensitive primary cells
- streamlined downstream processing, including dissolvable formats
Our product range includes P1 Synthetic, P2 Dissolvable, and X1 Dissolvable microcarriers, designed for applications across cell culture and cellular agriculture workflows.
As the field continues to scale, the choice of microcarrier system will play an increasingly important role in enabling efficient and reproducible bioprocesses.
If you’re interested in trying our microcarriers, feel free to contact us at info@smartmcs.com.au.