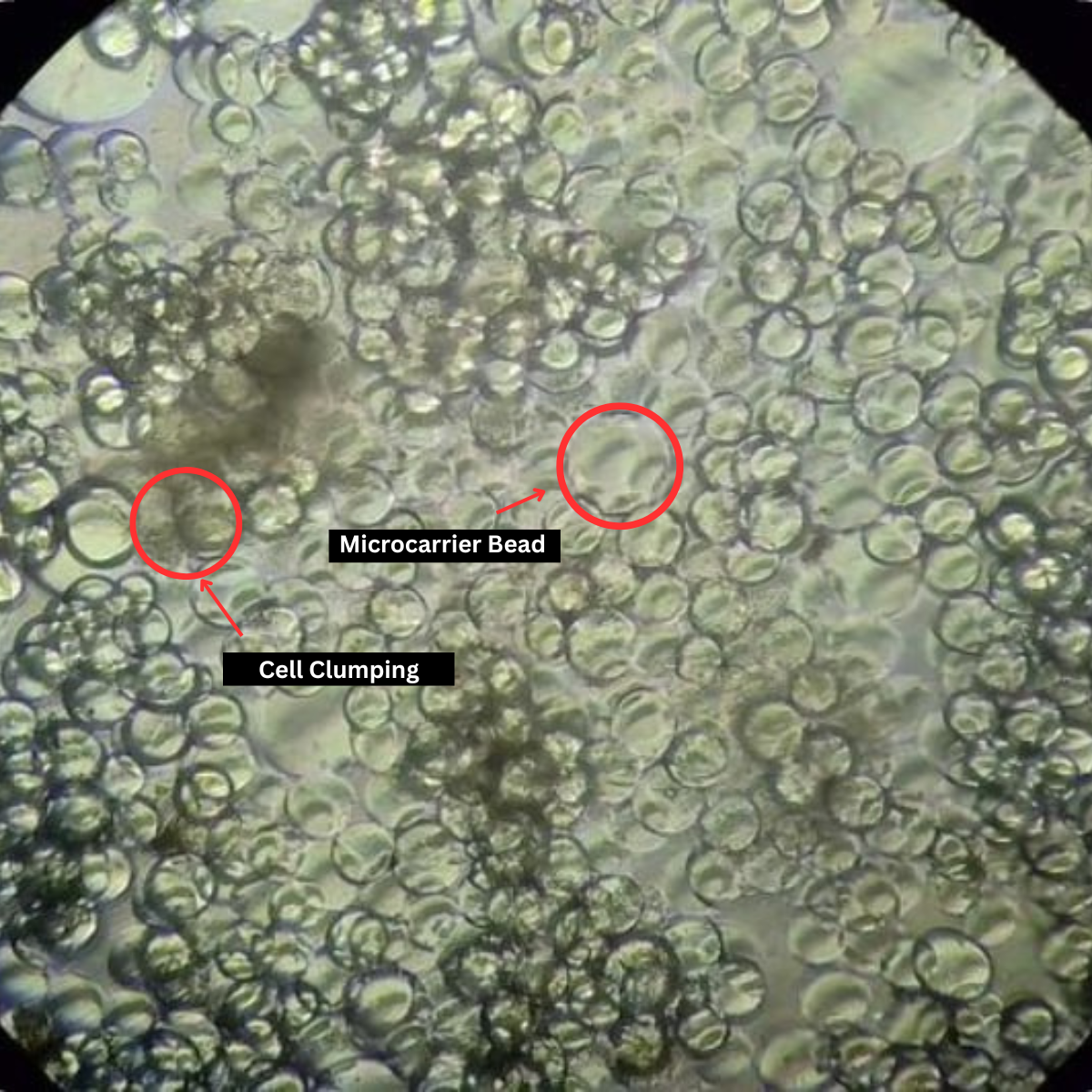
Microcarrier culture illustrating uneven cell seeding, where cells occupy some microcarriers and become confluent and clump, while others remain sparsely populated or empty.
“Why are my cells forming clumps instead of evenly coating microcarriers?”
Cell clumping is one of the most frequently encountered challenges in microcarrier-based culture, particularly when working with mesenchymal stromal cells (MSCs) and other highly adherent cell types.
Researchers often observe:
- Large cell–microcarrier aggregates
- Uneven cell distribution across the culture
- Reduced growth rates or viability
- Increased variability between runs
While some degree of aggregation can be expected as cells proliferate, excessive or uncontrolled clumping usually reflects process-related limitations rather than inherent cell behaviour.
What Drives Clumping in Microcarrier Systems?
Cell clumping rarely has a single cause. Instead, it emerges from the combined effects of cell properties, microcarrier characteristics, and hydrodynamic conditions.
Common contributing factors include:
- Uneven cell seeding across microcarriers, where some beads receive a high number of cells while others remain sparsely populated
- Insufficient agitation during the initial seeding phase, which can prevent uniform cell attachment and promote early aggregate formation
- Inappropriate microcarrier size or concentration, affecting collision frequency and the effective attachment surface available to cells
- Surface saturation during later culture stages, when microcarriers approach confluence and cells begin bridging between neighbouring beads
- Cell line–specific adhesion behaviour, as some cell types (including certain MSC lines) exhibit stronger cell–cell interactions and may therefore be more prone to aggregation
Identifying which of these factors is dominant in your system is key to controlling aggregation.
When Excessive Clumping Becomes a Problem
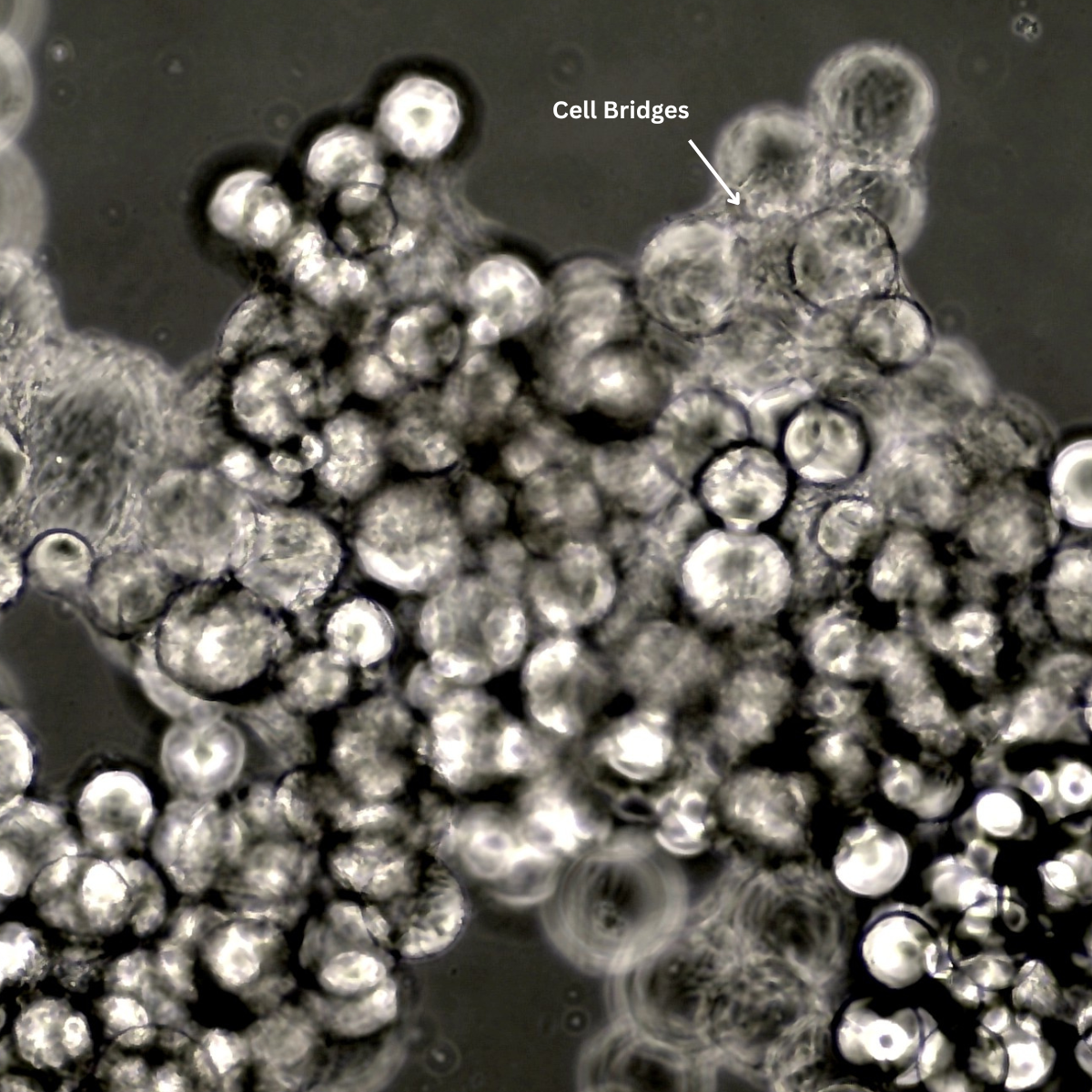
Micrograph showing cell bridges forming between neighbouring microcarrier beads as the culture reaches overall confluency and most microcarriers become occupied.
Excessive aggregation can compromise culture performance by:
- Limiting oxygen and nutrient transfer within dense aggregates
- Creating heterogeneous microenvironments that can reduce cell viability
- Resulting in inefficient use of available culture surface, thereby not maximising cell expansion and overall yield
- Altering cell behaviour due to changes in cell–cell signalling and mechanical environment
- Reducing productivity of cellular outputs such as secreted factors or extracellular vesicles
- Increasing variability during harvest
- Complicating downstream scale-up
Importantly, clumping itself is not inherently negative, but loss of control over aggregation reduces reproducibility, which is critical for both research and manufacturing workflows.
Practical Approaches to Reduce Clumping
1. Adjust Agitation for Uniform Suspension
Agitation is central to microcarrier culture, but optimisation is about consistency, not intensity.
- If agitation is too low, microcarriers settle and aggregate
- If agitation is too high, shear stress can damage cells
The objective is to maintain microcarriers in continuous, gentle motion without creating dead zones.
In practice, this often involves:
- Confirming microcarriers remain evenly suspended
- Increasing agitation gradually as cell mass increases
- Re-evaluating settings as culture progresses
2. Select an Appropriate Microcarrier Size and Density
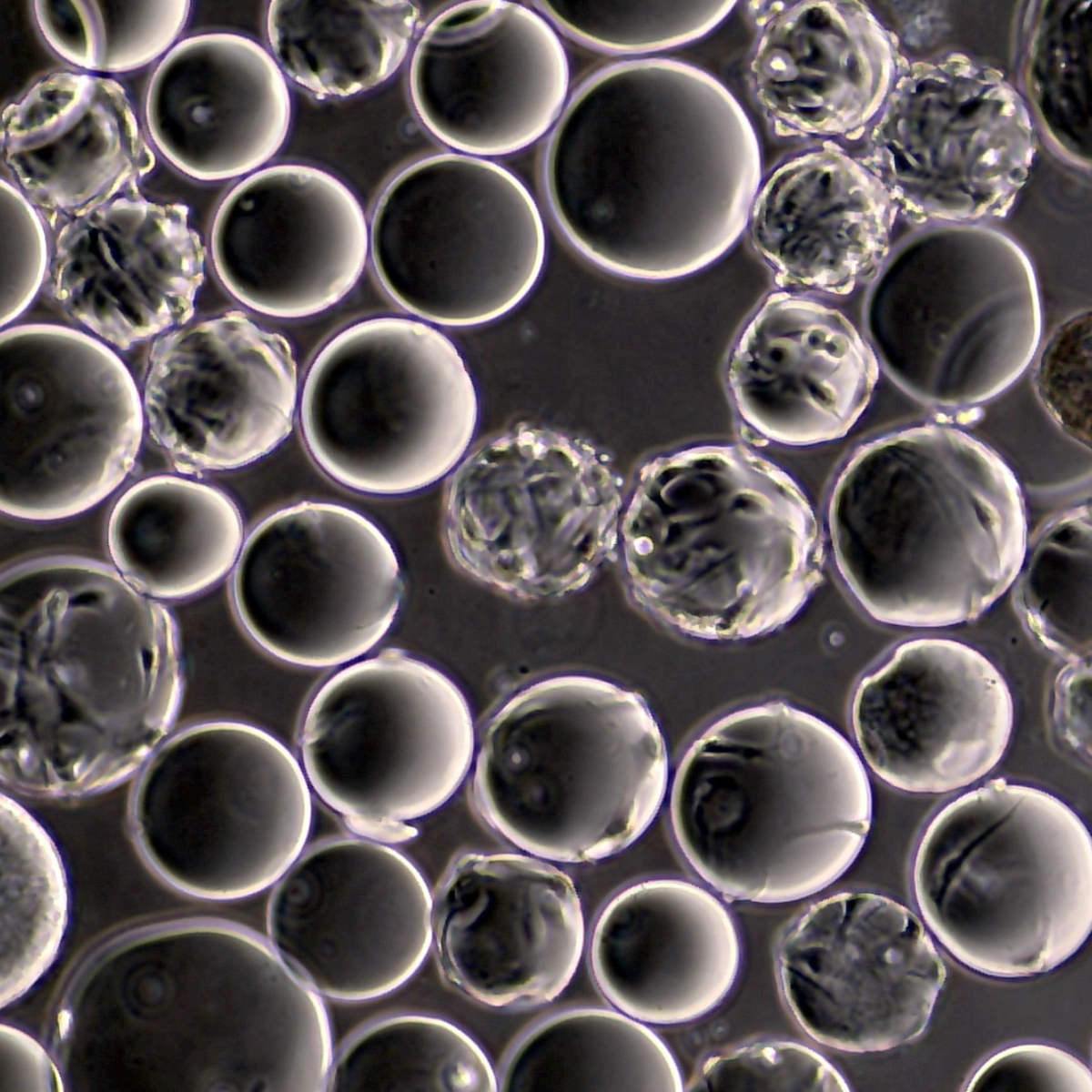
Micrograph showing healthy cell attachment across microcarriers.
Microcarrier size and surface density strongly influence aggregation behaviour.
Consider that:
- Smaller microcarriers increase collision frequency
- Larger microcarriers can reduce excessive aggregation but may limit total surface area and, if too large, may reduce cell attachment efficiency and increase the likelihood of cells experiencing shear stress.
- High microcarrier loading or using too many microcarriers in a given volume can promote local overgrowth and clump formation
Optimising these parameters requires balancing available surface area attachment efficiency, and manageable collision dynamics, particularly for MSC cultures.
3. Align Microcarrier Choice with Cell Type and Growth Phase
Clumping often increases as cultures approach high confluence.
This may occur when:
- Individual microcarriers become surface-saturated, and cells begin extending across neighbouring beads
- Cells start forming bridges between adjacent microcarriers, effectively “grabbing” and linking them into larger aggregates
- Cell–cell adhesion begins to dominate
- Cultures exceed optimal expansion windows
In these cases, increased aggregation may indicate that cells are ready for passaging or harvest rather than further expansion.
4. Use Anti-Clumping Agents Judiciously
Anti-clumping agents can help reduce excessive aggregation, especially during:
- High-density expansion
- Scale-up transitions
- Sensitive attachment phases
These agents act by moderating cell–cell interactions without compromising viability. However, they are most effective when combined with appropriate agitation and microcarrier selection, rather than used as standalone solutions.
Considerations During Scale-Up
Clumping often becomes more pronounced during scale-up due to:
- Larger working volumes
- More complex flow patterns
- Longer culture durations
Conditions that are stable in spinner flasks may not translate directly to bioreactors. Successful scale-up requires:
- Re-optimisation of agitation parameters
- Adjustment of microcarrier concentration
- Monitoring aggregation as a process variable rather than a visual artifact
A Practical Checkpoint
“Are cells interacting more with each other than with available microcarrier surface?”
If so, the solution likely lies in:
- Increasing effective surface availability (increase microcarrier usage / amount of microcarriers used)
- Improving mixing uniformity
- Adjusting culture timing
—not simply increasing agitation speed.
Key Points to Remember
- Cell clumping is common in microcarrier-based culture, particularly with MSCs
- Excessive aggregation usually reflects process imbalance
- Agitation, microcarrier size, and surface density must be aligned
- Anti-clumping agents are supportive tools, not primary fixes
- Controlled aggregation is manageable; uncontrolled aggregation limits scalability
Interested in Microcarriers for Cell Expansion?
For teams looking to optimise cell attachment, culture performance, and downstream processing, choosing the right microcarrier can make a significant difference.
These microcarriers are designed to support reliable cell attachment, scalable expansion, and efficient downstream recovery across a range of cell culture applications.
Final Thought
Microcarrier culture performance is shaped by the interaction between cell behaviour, surface availability, and mixing conditions. Rather than focusing on a single variable, consistent outcomes are more often achieved through balanced optimisation of these parameters.
By recognising early signs of aggregation and adjusting conditions methodically, researchers can maintain stable, reproducible microcarrier cultures that support reliable scale-up