“Why is microcarrier dissolution taking longer than expected?”
Microcarrier dissolution is a key step during harvest, but delays are usually linked to how the process is handled. In our experience, when the workflow is well understood and the steps are followed properly, dissolution is straightforward. When something isn’t quite right, or the process is still new, it can take longer than expected.
When dissolution does not go as planned, researchers often observe:
- Prolonged dissolution times
- Incomplete breakdown of microcarriers
- Delays during cell harvest
- Extended processing time during harvest and cell recovery
While some variation can occur, prolonged dissolution is often influenced by how the microcarriers were prepared earlier in the process.
What Drives Microcarrier Dissolution?

Cells recovered after P2 microcarrier dissolution using P2 Dissolving Buffer, supporting a simple harvest process with well-dispersed recovered cells.
Microcarrier dissolution rarely has a single cause. Depending on the microcarrier material, either enzymatic or non-enzymatic solutions may be used to achieve dissolution. However, the solution itself is only one part of the equation. Dissolution speed and efficiency are also shaped by several process parameters.
Common contributing factors include:
- The choice of dissolution solution (enzymatic or non-enzymatic)
- Agitation during dissolution
- Temperature during dissolution
- Washing efficiency prior to dissolution
- Microcarrier concentration relative to enzyme volume
- Incubation time
- Cell type and extracellular matrix production
Agitation can mechanically assist breakdown and improve contact between the dissolution solution and the microcarrier surface. Temperature should support enzyme activity while remaining appropriate for cell viability during harvest. Thorough washing helps remove residual proteins or storage components that may interfere with enzyme access. Higher microcarrier concentrations may require proportionally higher enzyme volumes to maintain effective activity. Incubation time must balance efficient dissolution with minimising cellular stress. In addition, cell types that produce substantial extracellular matrix can influence enzyme accessibility and overall dissolution kinetics.
Optimising these parameters supports efficient dissolution and consistent performance across runs.
When Slow Dissolution Becomes a Problem
Prolonged or uneven dissolution can directly affect harvest quality and cell recovery.
When dissolution takes longer than intended, cells may be exposed to enzymes or buffers for extended periods, which can reduce viability and increase cellular stress. If microcarriers dissolve incompletely, additional filtration or mechanical intervention may be required. In some cases, not all cells detach efficiently, leading to reduced total cell yield.
Incomplete dissolution may also leave residual particulates or microcarrier fragments in the recovered cell suspension. This can be especially problematic for clinical, translational, or sensitive downstream applications where product purity and quality control are critical.
Incomplete or variable dissolution can also introduce batch-to-batch variation, affecting the number of cells recovered and increasing overall process costs.
For these reasons, efficient dissolution is important not only for timing, but for maintaining cell quality, maximising recovery, reducing particulate burden, and ensuring predictable output.
Practical Approaches to Improve Dissolution
1. Pre-condition microcarriers before cell seeding
For dissolvable microcarriers, proper hydration and equilibration prior to use influence downstream dissolution behaviour.
Pre-conditioning microcarriers before cell seeding helps ensure they are fully hydrated under controlled conditions, supporting more efficient dissolution during harvest.
This step must always be completed before cell seeding and never with cells present.
2. Wash microcarriers thoroughly before use
Incomplete washing is a common cause of slow or uneven dissolution.
Residual storage components salts or proteins can:
- Interfere with dissolution by limiting enzyme access or altering reaction conditions
- Slow degradation kinetics
- Lead to variable dissolution rates between runs, inconsistent cell detachment, and differences in total cell recovery
Best practice includes:
- Multiple wash steps using an appropriate buffer, with sufficient soaking or equilibration time to allow full buffer exchange
- Ensuring removal of storage solution and residual culture supernatant or media components prior to dissolution.
Well-prepared microcarriers are easier to dissolve.
3. Balance time and temperature during dissolution
Dissolution behaviour is influenced by both temperature and exposure time. Temperature affects reaction kinetics and, in enzymatic systems, enzyme activity. Increasing temperature within an appropriate range can accelerate dissolution by increasing molecular motion and enzyme efficiency. However, temperature must remain compatible with cell viability.
Lower temperatures may slow dissolution kinetics and therefore require longer exposure times to achieve complete breakdown. The relationship is not linear, and optimal conditions depend on the specific system.
The appropriate balance depends on:
- The type of dissolution solution used
- The robustness of the cell type
- The desired recovery timeline
Some cell lines are relatively robust and tolerate a broader range of conditions. In contrast, more sensitive cell types, such as induced pluripotent stem cells (iPSCs), may require tighter control of temperature and exposure time to minimise stress.
Faster dissolution is not inherently better. Conditions should be optimised to support efficient carrier breakdown while maintaining cell viability and recovery quality.
4. Use the right amount of dissolution buffer
Using too many microcarriers with too little dissolution buffer can slow down dissolution.
This can lead to:
- Slower breakdown
- Uneven dissolution
- Partly dissolved microcarriers
- More leftover particles
Always follow the recommended protocol and start with the suggested microcarrier-to-buffer ratio. If the microcarriers still do not dissolve well, you may need to carefully increase the amount of dissolution buffer while checking that the cells remain healthy.
Common Causes of Slow Dissolution
In practice, slow dissolution is often linked to washing, buffer volume, temperature, or mixing.
- Not enough washing beforehand
- Too little dissolution buffer for the amount of microcarriers
- Temperature not suitable for enzyme activity
- Poor mixing during the harvest step
If breakdown is slow or incomplete, review these steps first before changing the overall workflow. Small adjustments to washing, buffer volume, temperature, or mixing can often improve the result.
A Practical Checkpoint
If the answer is no, the solution lies in preparation rather than in the dissolution step itself.
Key Points to Remember
- Microcarrier dissolution is a critical process step
- Enzyme concentration must match microcarrier load
- Temperature and mixing influence reaction kinetics
- Conditions should be optimised for the specific cell type
Questions Researchers Commonly Ask
A Practical Solution: P2 Dissolving Buffer
For researchers using P2 dissolvable microcarriers, P2 Dissolving Buffer can help speed up the harvest process. It is designed to work together with P2 microcarriers and TrypLE to support faster and more controlled microcarrier dissolution.
Under optimised conditions, dissolution and cell recovery can typically be completed in around <10 minutes. This can help reduce the time cells are exposed during harvest, limit extra handling steps, reduce the need for filtration or mechanical separation, and support reliable cell recovery while maintaining good cell viability.
P2 Dissolving Buffer should be used with P2 dissolvable microcarriers and according to the recommended protocol.
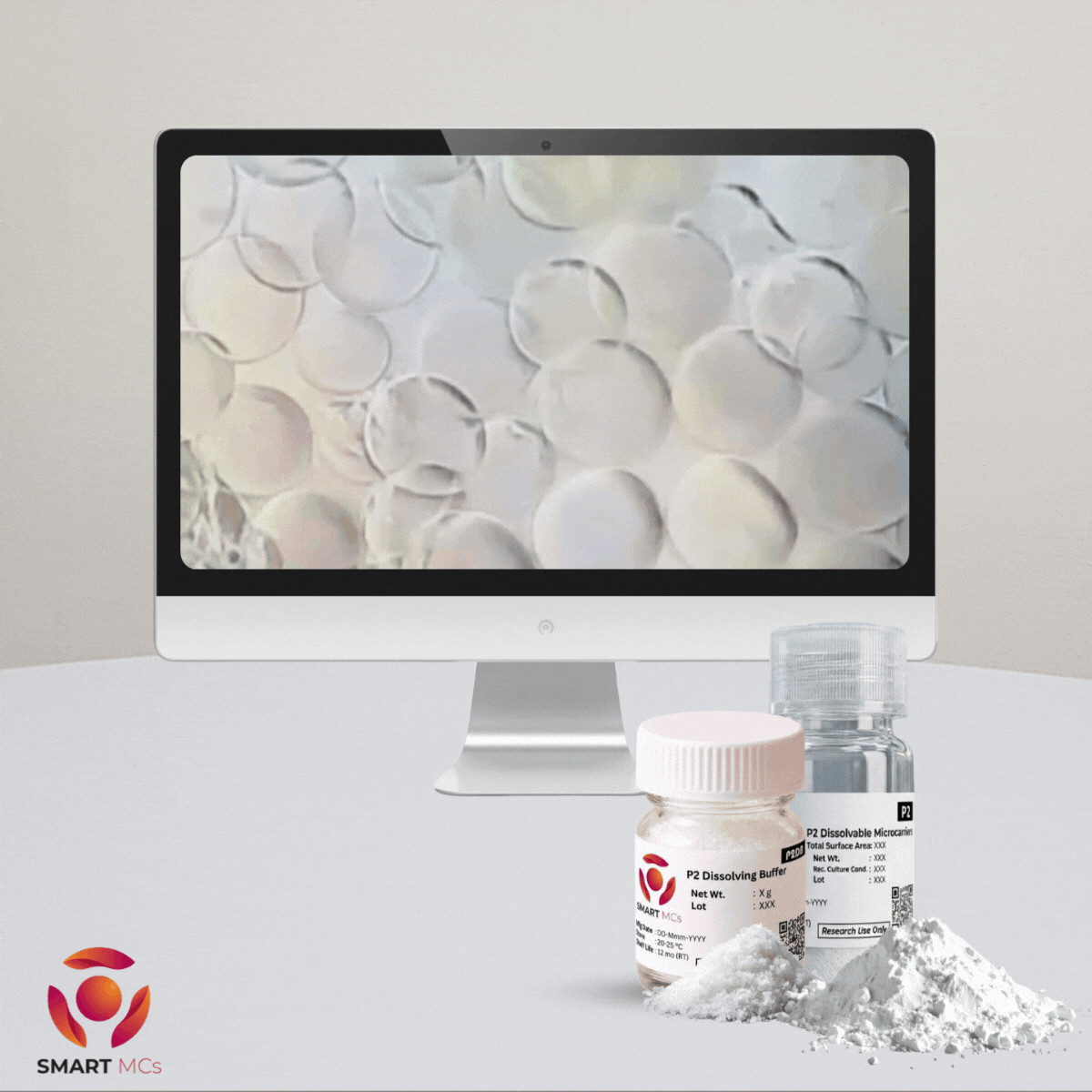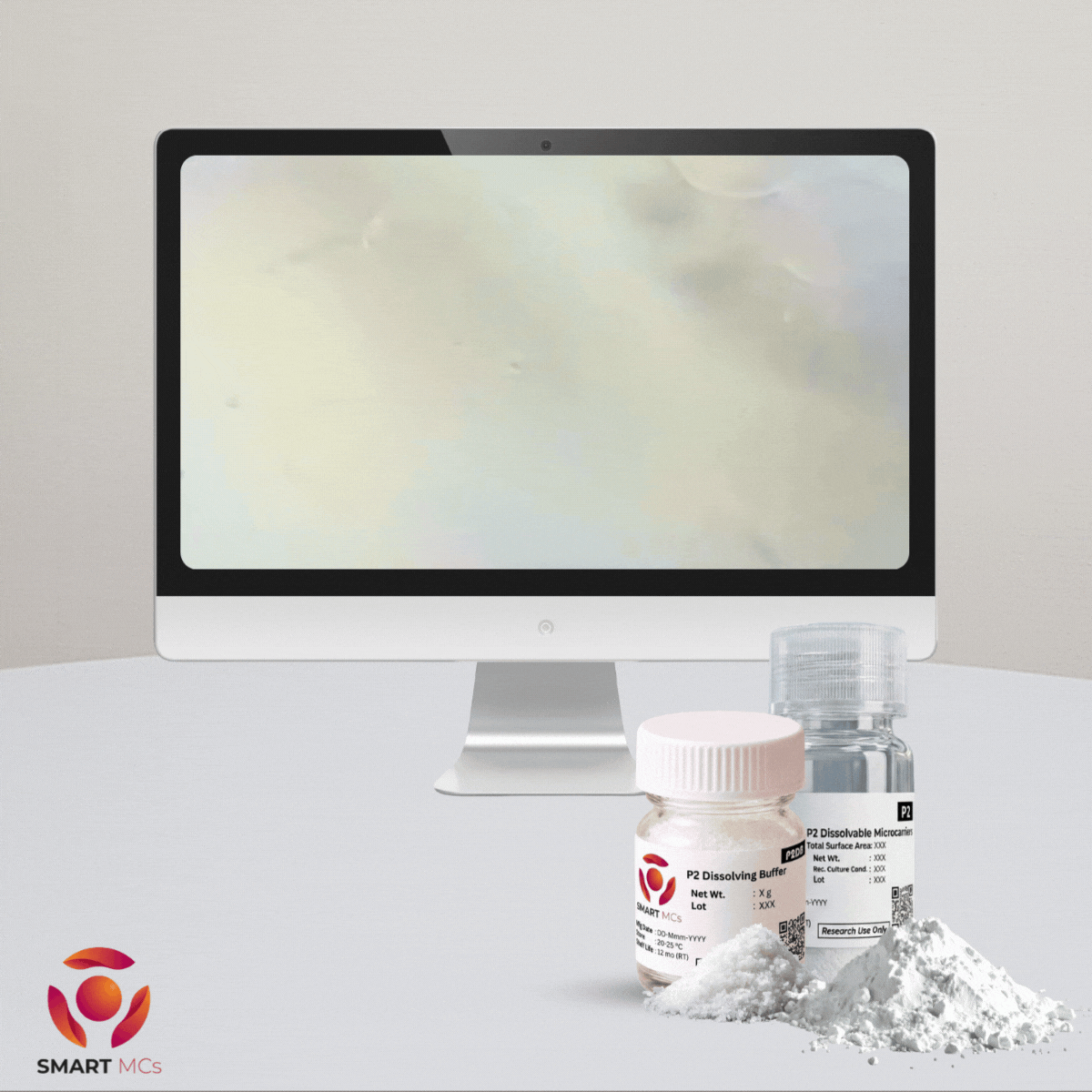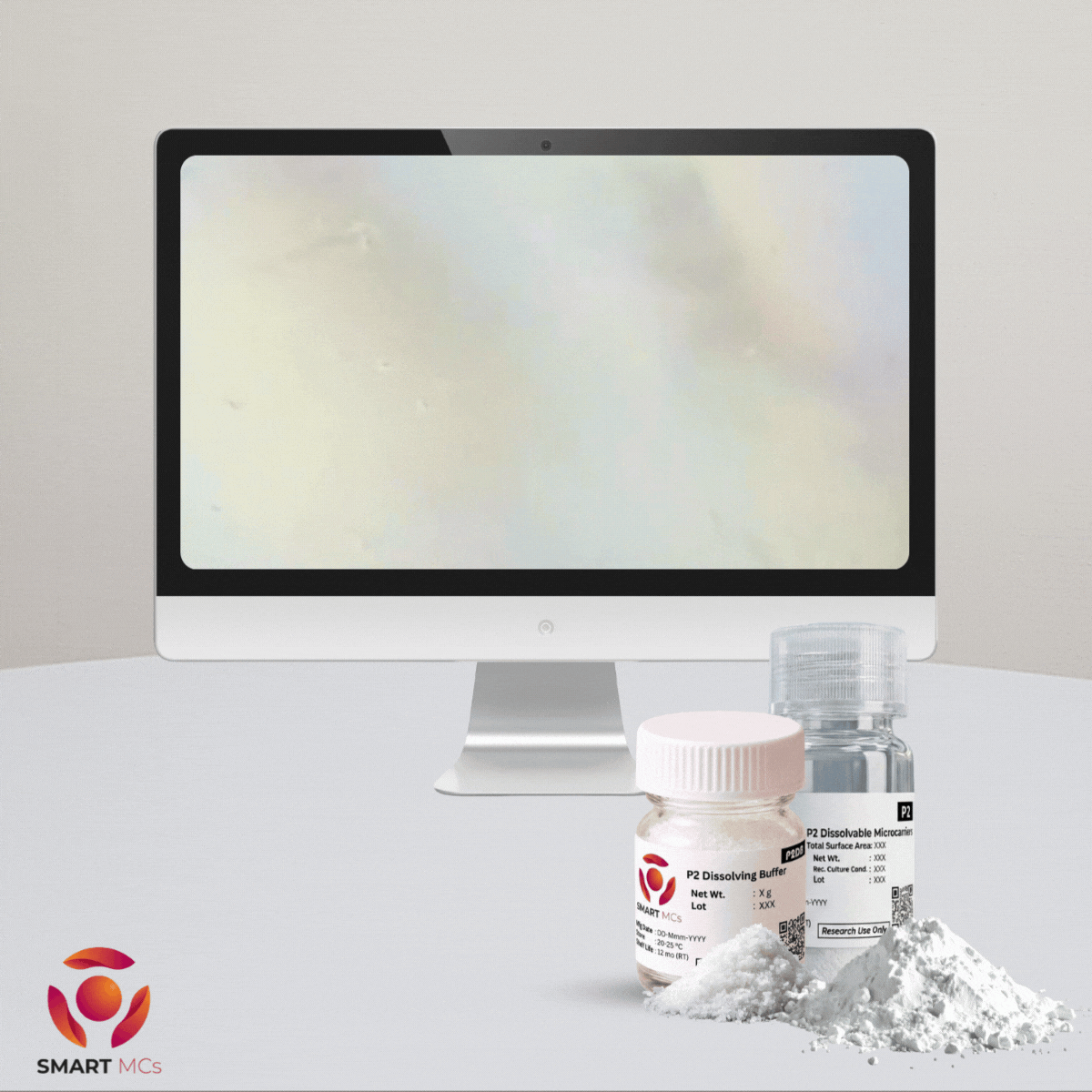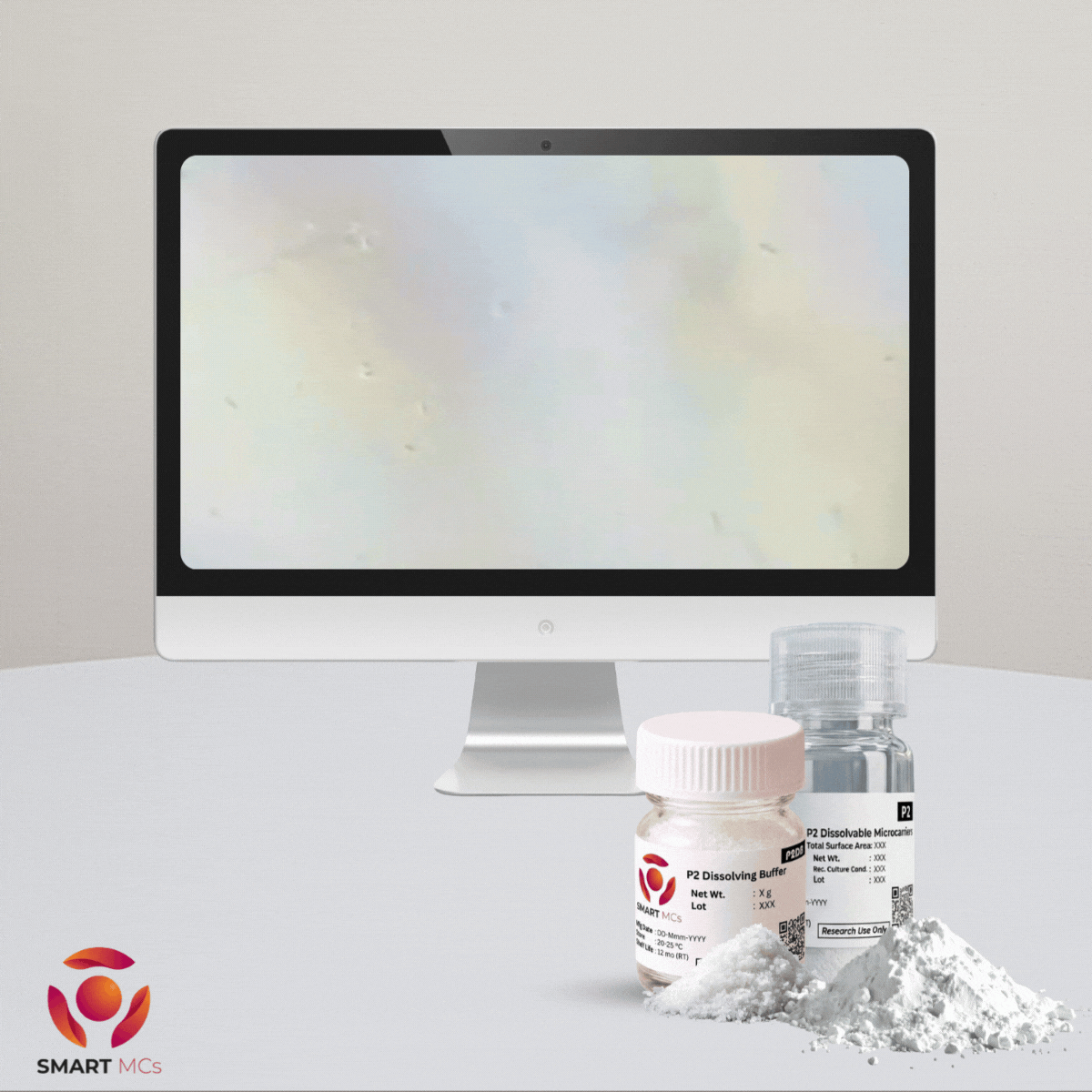
Product Spotlight: P2 Microcarriers and P2 Dissolving Buffer work together with TrypLE to support rapid microcarrier dissolution and cell recovery in <10 minutes under optimised conditions.